C5TR01
Viva CRT-P
Device Survival Probability
C5TR01_C6TR01_SURV
Loading...
| 1 yr | 2 yr | 3 yr | 4 yr | 5 yr | 6 yr | 7 yr | 8 yr | at 97.0 mo | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| % | 100.0 | 100.0 | 100.0 | 99.9 | 99.9 | 99.9 | 99.9 | 99.8 | 99.8 | ||
| % | 99.9 | 99.7 | 99.3 | 97.5 | 95.1 | 90.8 | 84.1 | 64.7 | 59.8 | ||
| # | 7364 | 6603 | 5917 | 5148 | 4408 | 3633 | 2422 | 359 | 210 |
- Including Normal Battery Depletion – This curve includes devices that have reached at least 80% of expected longevity. This curve is most representative of clinical performance and how long the device will last.
- Excluding Normal Battery Depletion – This is the malfunction free survival curve.
Product Characteristics
| Generator Type | CRT-Pacing |
|---|---|
| NBD Code | OAE-DDDR |
| Max. Delivered Energy | N/A |
| Connector Style | CRT-P Style |
|---|---|
| Serial Number Prefix | PZZ |
| Mass | 26 g |
| Volume | 15 cc |
| Waveform | N/A |
| X-Ray ID | PVX |
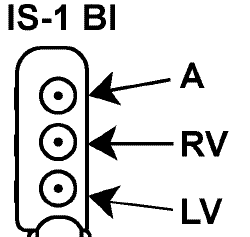
CRT-P Style
Estimated Longevity
| Amplitude Setting | 500 Lead Ω | 1000 Lead Ω |
|---|---|---|
| Low 2.5 V (A, RV) | 8.5 | 10.4 |
| Nominal 3.5 V (A, RV) | 5.8 | 8.0 |
| High 5.0 V (A, RV) | 3.2 | 5.0 |
Longevity estimates based on the following device usage. Pace/Sense Mode DDD ; Atrial Pulse Width 0.4 ms; Right Ventricle Pulse Width 0.4 ms; Lower Pace Rate 60 bpm; Right Ventricle Percent Paced 100 %; Upper Sensing Rate 130 ppm; Atrial Percent Paced 100 %; Left Ventricle Pulse Width 0.4 ms; Left Ventricle Pulse Amplitude same as A, RV; Left Ventricle Percent Paced 100 %; Left Ventricle Lead Impedance same as A, RV; EGM Prestorage ON ; EGM Prestorage ON Time 1 ; Atrial Fib Percent of Time 0 %Hide this content
Distribution Data
| US Market Release | |
|---|---|
| CE Approval Date | 2014-04-04 |
| Registered USA Implants | |
| Estimated Active USA Implants | |
| Normal Battery Depletions |
| US Market Release | |
|---|---|
| CE Approval Date | 2014-04-04 |
| Estimated WW Distribution | 12766 |
| Normal Battery Depletions | 4 |
EOS Indication
From the point that the RRT is set, the pacemaker will operate for approximately three months for typical pacemaker configurations during the normal operating life.
Data as of April 30, 2024