D164VWC
Virtuoso VR
Device Survival Probability
D154VWC,D164VWC_SURV
Loading...
| 1 yr | 2 yr | 3 yr | 4 yr | 5 yr | 6 yr | 7 yr | 8 yr | 9 yr | 10 yr | 11 yr | 12 yr | 13 yr | 14 yr | 15 yr | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % | 100.0 | 100.0 | 99.9 | 99.7 | 98.1 | 96.8 | 96.7 | 96.6 | 96.5 | 96.5 | 96.4 | 96.4 | 96.4 | 96.4 | 96.3 | |||||||
| % | 99.8 | 99.6 | 99.3 | 98.8 | 96.0 | 87.7 | 77.5 | 60.3 | 44.4 | 31.9 | 22.8 | 18.0 | 16.3 | 15.2 | 14.2 | |||||||
| # | 28535 | 26124 | 23730 | 21531 | 19161 | 16195 | 13286 | 9329 | 6103 | 3950 | 2478 | 1605 | 1153 | 849 | 126 |
- Including Normal Battery Depletion – This curve includes devices that have reached at least 80% of expected longevity. This curve is most representative of clinical performance and how long the device will last.
- Excluding Normal Battery Depletion – This is the malfunction free survival curve.
Product Characteristics
| Generator Type | ICD |
|---|---|
| NBD Code | VVE-VVIR |
| Max. Delivered Energy | 35 J |
| Connector Style | Cx Style |
|---|---|
| Serial Number Prefix | PUP |
| Mass | 68 g |
| Volume | 37 cc |
| Waveform | Biphasic |
| X-Ray ID | PVR |
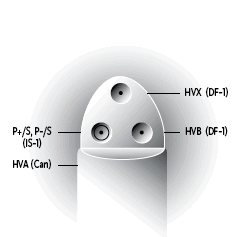
Cx Style
Estimated Longevity
| Charging Frequency | 100% Pacing | 50% Pacing | 15% Pacing | 100% Sensing |
|---|---|---|---|---|
| Monthly | 4.8 | 5.1 | 5.3 | 5.4 |
| Quarterly | 8.1 | 9.0 | 9.6 | 10.0 |
| Semi-annual | 10.0 | 11.2 | 12.3 | 12.9 |
Longevity estimates based on the following device usage. Pacing Mode VVI; Ventricular Lower Pacing Rate 60ppm; Ventricular Pulse Width 0.4ms; Ventricular Pulse Amplitude 3V; Ventricle Impedance 510ohms; Ventricle Sensing Rate 70bpm; EGM prestorage OFF; Optivol ON; Charging frequency assumes maximum energy charge and includes therapy shocks and capacitor formation. Hide this content
Distribution Data
| US Market Release | |
|---|---|
| CE Approval Date | 2006-03-07 |
| Registered USA Implants | 1 |
| Estimated Active USA Implants | 1 |
| Normal Battery Depletions |
| US Market Release | |
|---|---|
| CE Approval Date | 2006-03-07 |
| Estimated WW Distribution | 14374 |
| Normal Battery Depletions | 268 |
EOS Indication
EOS device status is defined as 3 months following an RRT indication assuming the following PSP conditions: 100% VVI pacing at 60 min-1, 2.5 V, 0.4 ms; 500 Ohm pacing load; and 6 full-energy charges. EOS may be indicated before the end of 3 months if the device exceeds these conditions.
Data as of December 1, 2023